The origin of air-breathing was one of the great moments in the history of vertebrate evolution. Air-breathing was an important adaptation allowing vertebrates to invade terrestrial environments and air-breathing among the fishes is of interest, in part, due to its relatedness to the transition of the vertebrates from an aquatic to a terrestrial existence. This adaptation, however, and others such as urea utilization (This guy Griffith wrote a very good read of this in Bioscience in 1991) and the development of fins with more bony structure and musculature, shortly to become legs, occurred early within the fishes and has a long history of use among the fishes before, during and after the rise of terrestrial vertebrates.
The diversity of modification among fishes and the link to terrestrial movement that some of these modifications suggest have been the basis of the instructiveness of creating scenarios as a means to advance knowledge in an area of high speculation.
Here is a short scenario of air-breathing among fishes and links to terrestrial emergence that ignores, for the most part, fishes that are in a more direct line to the first terrestrial vertebrates, showing that these adaptations came about and persist in fully aquatic organisms.
Breathing Essentials1) Early air-breathing organs.
Primitive fishes among many lines, such as the Reedfish – considered the primitive ray finned fish – have air-breathing organs and the air blabber is thought to have existed early in vertebrate evolution, at least by the Silurian. The first air filled organ was probably used as a lung and the use of an air filled sac for buoyancy was derived later. Although the structure used for air-breathing differs in modern fishes many bony fishes (teleosts) utilize air-breathing (49 families and nearly 400 species).
2) Chemosensitivity:
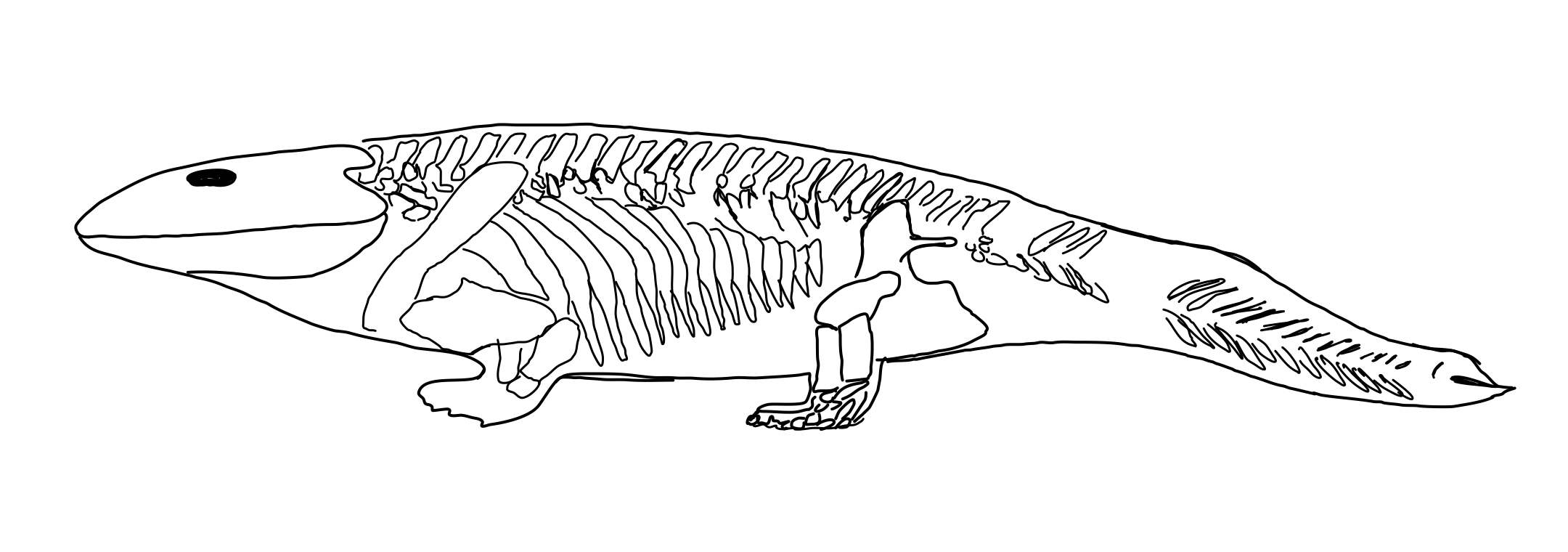
Neural control of lung ventilation – based on the presence of CO2– which exists in the tetrapods and has been shown in gars. This suggests to the authors that a common air-breathing ancestor of actinopterygians (ray-finned) and sarcopterygians (lobed-finned--with bones extending into the fins) existed prior to divergence of these two lines. This modification also likely occurs in more modern fishes that breathe air. Some features that gars have are also seen in the ‘fishapod’ fossil Tiktaalik (Figure 1).
3) Air Exchange/Innervation:
Among teleosts that use an air-breathing organ most ventilate via a buccal pulse pump. This is also found in lungfishes, frogs, and salamanders. The neural and muscle control of the pump in bony fishes is identical to that in lungfishes, frogs, and salamanders. Due to the commonality of this pattern it likely existed early in vertebrate evolution before tetrapod divergence.
4) Pulmonary Surfactant:
Pulmonary surfactant is a mixture of lipids and proteins which reduce surface tension at air-liquid interface. The substance is necessary to allow air-breathing. This substance exists in all major vertebrate groups including the fishes and is present even in fish who currently do not breath air such as goldfish . This finding, as with the modifications above, suggest an early and common origin among vertebrates.
Why did fish develop air-breathing?
One scenario for the origin of air-breathing is related to hypoxic conditions in shallow water. Under this hypothesis hypoxic conditions, brought about by drying or stagnant pools or similar situations, would make air-breathing beneficial. A similar hypothesis exists for the advent of urea utilization among fishes, limited water flow in pools or near shore environments with limited water exchange could cause toxic ammonia buildup making ammonia to urea conversion beneficial. Alternative hypotheses to the origin of air-breathing do exist. One such hypothesis is that the advent of air-breathing increased the amount of oxygenated blood entering the heart. The air-breather under this scenario would gain the advantage of well oxygenated cardiac muscle thus allowing extended activity.
Up for debate:
Most fossils of earlier tetrapods and fishes near to amphibious in nature (Acanthostega, Icthyostega, Pandericthys) have been found in freshwater deposits. Ichthyostega is a, mostly terrestrial dwelling, amphibian, but it still retains a fishy tail (Figure 2).
Therefore, it has been suggested that the origin of tetrapods occurred in a freshwater environment. Since some fishes with ancient origins, such as the coelacanth and elasmobranchs, utilize urea as a osmoregulatory device in marine waters and thus it has also been suggested that the origin of urea use and perhaps, therefore, the progenitors of tetrapods, lived in a marine environment. Also up for debate is the single origin of urea utilization; a single origin, before elasmobranch divergence, or two origins, once within the elasmobranch line and again before Sarcopterygian divergence along with the breathing oriented modifications. (okay, a quick aside, this subject needs to be dealt with in more depth for a full understanding, and I am going to cheap out on you and skip most of it--maybe I should cover this more in-depth in another post).
Urea use, leg/fin muscle development, air-breathing and the related modifications outlined above occurred early in the evolution of vertebrates and exist within the fishes as well as the tetrapods. It would seem fitting to use fishes as models to suggest what may have occurred.
The Scenario: Behavior changes first:
Although several environmental challenges may have led to the retention of these modifications, my suggested scenario is that fishes, to increase fitness, started to seek out secluded/protected areas in near shore environments to deposit eggs. It doesn’t matter whether the habitat of adult fish is freshwater or marine as long as movement inshore for developmental purposes takes places. Of course many levels of progression can be imagined – the first to be inhabited were tidal pools or pools formed by receding waters to pools entirely disconnected from larger water bodies.
I suggest that for this behavioral seeking to take place none of the modifications outlined above were required. Once the behavior existed then adaptive pressures would exist that allowed modifications to be retained. Once the majority of modifications existed it would be a short step (intended) to a wholly terrestrial existence. I’ve outlined below models from extant fishes in a progressive nature to illustrate the possibilities of this scenario. I didn’t include lungfishes on purpose even though lungfishes are considered the closest extant cousins to tetrapods and are related to Rhipidistian fishes, which are the likely terrestrial amphibian’s ancestors. The adaptations that allowed eventually leaping out on to land all seem to come about in fishes and that still exist there.
Fish models and levels of emergence
A) The three-spined stickleback Gasterosteus aculeatus(an estuarine species) spawns in streams or into tidal pools high in the intertidal zone. Its physiology allows it to survive in these near-shore shallow habitats.
B) Among marine fish a model is found with toadfish. Toadfish, marine bottom dwellers, move inshore in order to spawn and nest under rocks and other submerged objects to which they attach their egg. Developing toadfish are also confined to the nests during development. Toadfish have been found to utilize urea (converting to it from ammonia) which may be beneficial to prevent the adult toadfish from poisoning their young in the next with the more toxic ammonia.
C) If urea use is beneficial during development – to prevent toxic build up of ammonia within a confined nest (and water) situation perhaps – then we would expect to see it in other fishes and we do. The guppy and trout, which do not excrete urea as adults excrete urea during development..
D) Mudskippers are amphibious fish that spend much of their time out of water. These fish breathe air, utilizes fin muscularization to crawl about, and also excrete urea. Mudskippers are awesome little mangrove inhibitors (Figure 3a and 3b).
E) A freshwater model of development within pools occurs in the African catfish Clarias gariepinus. Clarias, which during early development are exposed to pools with diminishing water also excrete urea. Clariasis also an air-breather and can move about on land. Clariascompletes our possible connections of modifications.
F) One further note that allows us to reach a pinnacle of behavior concerns the characin Brycon petrosus. These fish were observed moving onto land and spawning amongst vegetation. This takes our Clarias model one step further at least behaviorally. Breading inshore and finally out of the water completely, was probably the major impetus for terrestrial living among vetebrates.
The processes that led to terrestrial existence came about from a behavioral change to protect eggs and young by first moving to shallow water to lay eggs, stepping then to finding secluded pools, to eventually laying eggs completely out of the water. Other modifications that increased fitness (such as air-breathing and urea use), came later in the process, as fish moved higher, and/or spent more time in shallow water environments. A behavioral progression took place buoyed by physiological and anatomical adaptations. As the need arose to get further from predators the modifications that allowed such movement (and survival during out of water periods) would be selected. And conversely the more of these modifications that existed the better the fish would be at finding secluded pools in which to lay their eggs and the better the protection would be from predators. Damn this evolution stuff is fun.
Readings, more extensive than usual.
Ahlberg, P. E. and A. R. Milner 1994. The origin and early diversification of tetrapods. Nature 368: 507-514.
Brainerd, E. I., J. S. Ditelberg and D. M. Bramble 1993. Lung ventilation in salamanders and the evolution of vertebrate air-breathing mechanism. Biol. J. Linn. Soc. 49: 163-183.
Bruton, M. N. 1979. The survival of habitat desiccation by air breathing clariid catfishes. Env. Biol. Fish. 4: 273-280.
Farrell, A. P. and D. J. Randall 1978. Air-breathing mechanics in two Amazonian teleost,Arapaima gigasand Hoplerytheinus unitaeniatus. Can. J. Zool. 56:
Gans, C. 1985. Scenarios: Why? In: Evolutionary Biology of Primitive Fishes. Foreman, R.E. Gorbman, A. Dodd, J.M. and Olsson, R (eds). New York, Plenum Press. 1-9.
Graham, J. B. 1997. Air-breathing fishes: evolution, diversity, and adaptation. San Diego, Academic Press.
Greenwood, P. H. 1955. Reproduction in the catfish, Clarias mossambicus Peters. Nature, Lond. 176: 516-518.
Gregory, R. B. 1977. Synthesis and total excretion of waste nitrogen by fish of the Periophthalmus(mudskipper) and Scartelaosfamilies. Comp. Biochem. Physiol. 57A: 33-36.
Griffith, R. W. 1991. Guppies, toadfish, lungfish, coelacanths and frogs: a scenario for the evolution of urea retention in fishes. Env. Biol. Fish. 32: 199-218.
Hedges, S. B., C. A. Hass and L. R. Maxson 1993. Relations of fish and tetrapods. Nature 363: 501-502.
Kramer, D. L. 1978. Terrestrial group spawning of Brycon petrosus(Pisces: Characidae) in Panama. Copeia 3: 536-537.
Liem, K. F. 1988. Form and function of lungs: the evolution of air breathing mechanisms. Amer. Zool. 28: 739-759.
Neyt, C., K. Jagla, B. Thisse, L. Haines and P. D. Currie 2000. Evolutionary origins of vertebrate appendicular muscle. Nature 408: 82-86.
Neil H. Shubin, Edward B. Daeschler and Farish A. Jenkins, Jr. 2006. The pectoral fin of Tiktaalik roseae and the origin of the tetrapod limb. Nature. 440 (7085): 764–771.
Sullivan, L. C., C. B. Daniels, I. D. Phillips, S. Orgeig and J. A. Whitsett 1998. Conservation of surfactant protein A: evidence for a single origin for vertebrate pulmonary surfactant. J. Mol. Evol. 46: 131-8.
Terjensen, B. F., J. Verreth and F. H.J. 1997. Urea and ammonia excretion by embryos and larvae of the African catfish Claria gariepinus(Burchell 1822). Fish Physiol. Biochem. 16: 311-321.
Thompson, K.S. 1967. Notes of the relationships of the Rhipidistian fishes and the ancestry of the tetrapods. J. Paleontology. 41 (3): 660-674.
Wilson, R. J. A., M. B. Harris, J. E. Remmers and S. F. Perry 2000. Evolution of air-breathing and central CO2/H+ respiratory chemosensitivity: new insights from an old fish? The J. Exp. Biol. 203(22): 3503-3512.



 RSS Feed
RSS Feed